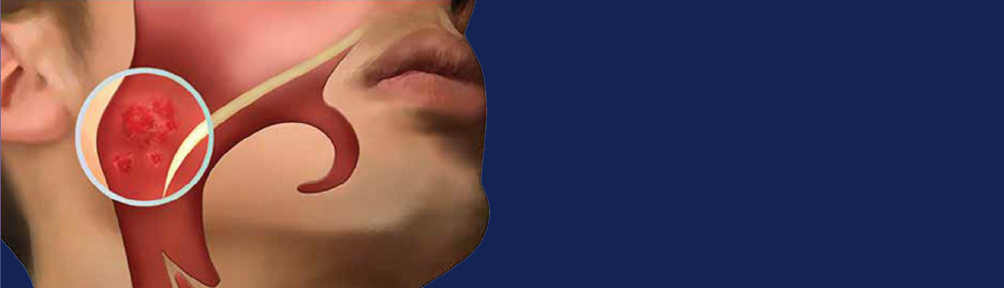
Nasopharyngeal Carcinoma
Recombinant Antigen-Antibody Detection
 Restalyst NPC-REAAD EBV EA-IgA ELISA is an in-vitro diagnostic intended for the qualitative and semi-quantitative detection of IgA antibodies in human serum/plasma. The test utilises specific proteins of Epstein-Barr virus (EBV) to aid in the diagnosis of nasopha- ryngeal carcinoma (NPC) by detecting the patient’s EA (Early Antigen) IgA antibody response in serum/plasma. These highly specific EBV proteins, generated using DNA recombinant technology, are known to be associated with nasopharyngeal carcinoma.
Restalyst NPC-REAAD EBV EA-IgA ELISA is an in-vitro diagnostic intended for the qualitative and semi-quantitative detection of IgA antibodies in human serum/plasma. The test utilises specific proteins of Epstein-Barr virus (EBV) to aid in the diagnosis of nasopha- ryngeal carcinoma (NPC) by detecting the patient’s EA (Early Antigen) IgA antibody response in serum/plasma. These highly specific EBV proteins, generated using DNA recombinant technology, are known to be associated with nasopharyngeal carcinoma.
NPC-REAAD determines the patients’ risk level of nasopharyngeal carcinoma, either positive or negative, by using an arbitrary unit of measurement known as REAAD-units (RU).
Recombinant NPC antigens are coated on microwells. Specific antibodies (human IgA against EBV EA proteins) will bind with the immobilised antigens, forming antigen-anti- body complexes. Excess antibodies are removed through subsequent washing steps. Secondary antibodies conjugated with horseradish peroxidase (HRP) are added to bind to the complexes. A substrate solution (TMB) is added and is converted by the enzyme to a detectable form (color signal). The intensity of this colored product is directly proportional to the concentration of antigen present in the original specimen. Microwells can be read on any suitable spectrophotometer or ELISA reader.